 Late last year, Australian stem cell and regenerative medicine company, Cynata Therapeutics Limited (ASX: CYP), developed compelling data in an asthma model and is now ramping up that program.
Late last year, Australian stem cell and regenerative medicine company, Cynata Therapeutics Limited (ASX: CYP), developed compelling data in an asthma model and is now ramping up that program.
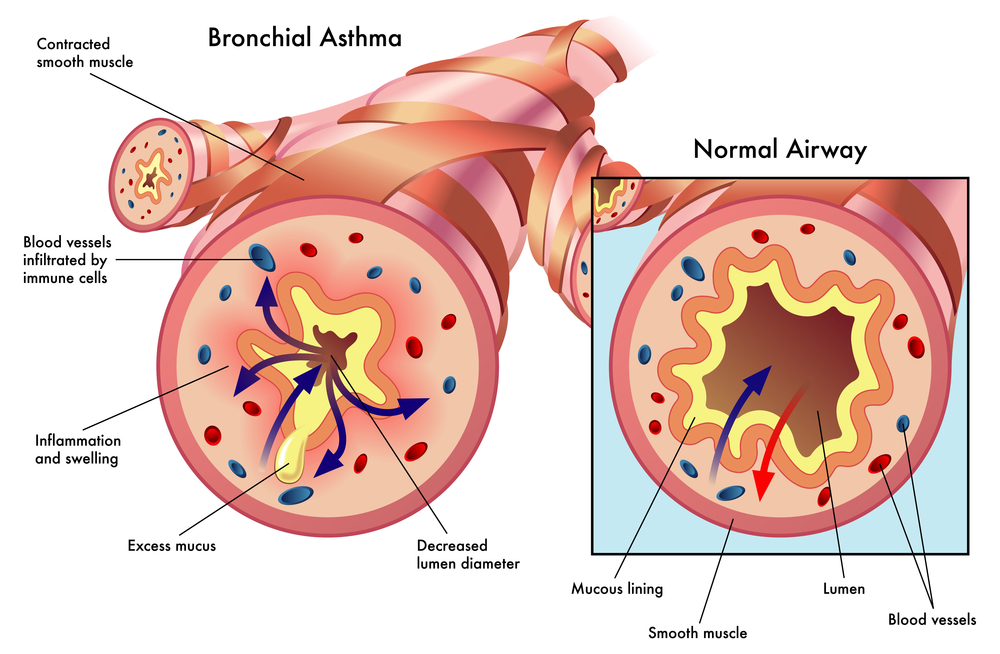
In October 2016, Cynata announced it had received convincing data from a proof of concept study of its Cymerus™ iPSC-derived mesenchymal stem cells (MSCs) in an experimental model of asthma. Those initial results demonstrated that Cymerus™ MSCs can exert a powerful effect via improved airway hyper-responsiveness.
More recently, on March 2, 2017, Cynata signed an agreement with the Monash Lung Biology Network, a consortia involving researchers from the Monash Biomedicine Discovery Institute and Department of Pharmacology at Monash University in Melbourne to execute a further preclinical study supporting the use of Cymerus™ MSCs for the treatment of asthma.
Cynata made that announcement after receiving the final report of a study which demonstrated that Cymerus™ MSCs also consistently reduced markers of airway inflammation and airway remodeling.
The final report of the initial study at Monash presents evidence that Cymerus™ MSCs could exert disease-modifying effects in asthma, which would revolutionize treatment approaches to the disease, as current treatments address symptoms and not underlying causes. Currently, the American Academy of Allergy, Asthma, and Immunology estimates that over 300 million people worldwide suffer from asthma, with 250,000 annual deaths attributed to the disease and the number of people affected by asthma anticipated to grow by more than 100 million by 2025.
Because of these promising preclinical finding and the company’s recent capital raising of A$6.0 million, Cynata will now conduct a further study to focus on the effects of Cymerus™ MSCs in combination with and in comparison to corticosteroids, the most common type of drugs used to control and prevent asthma symptoms.
Fujifilm also took a recent A$3.97 million strategic equity stake in Cynata. This further positions the company to pursue both the asthma program and its upcoming clinical trial investigating its lead Cymerus™ product CYP-001 for GvHD, which represents the world’s first clinical trial involving an allogeneic iPSC-derived MSC product.
To learn more about Cynata’s involvement with preclinical asthma research and its potential to revolutionize this disease indication, view Cynata’s recent announcement, titled “Cynata Advances Development of Cymerus™ MSCs for the Treatment of Asthma.”



















Tell Us What You Think!