Advanced therapy companies have been rapidly populating over the past few years, making it a high-value, fast-growth industry. Key drivers include high rates of clinical trials, accelerated pathways for product approvals, new technologies to support cell and gene therapy manufacturing, and the potential for advanced therapies to revolutionize healthcare.
Stem Cell Industry Growth
Stem cell treatments are one of several segments that compose the advanced therapies industry, which includes but is not limited to cell therapy, gene therapy, and tissue engineering applications.
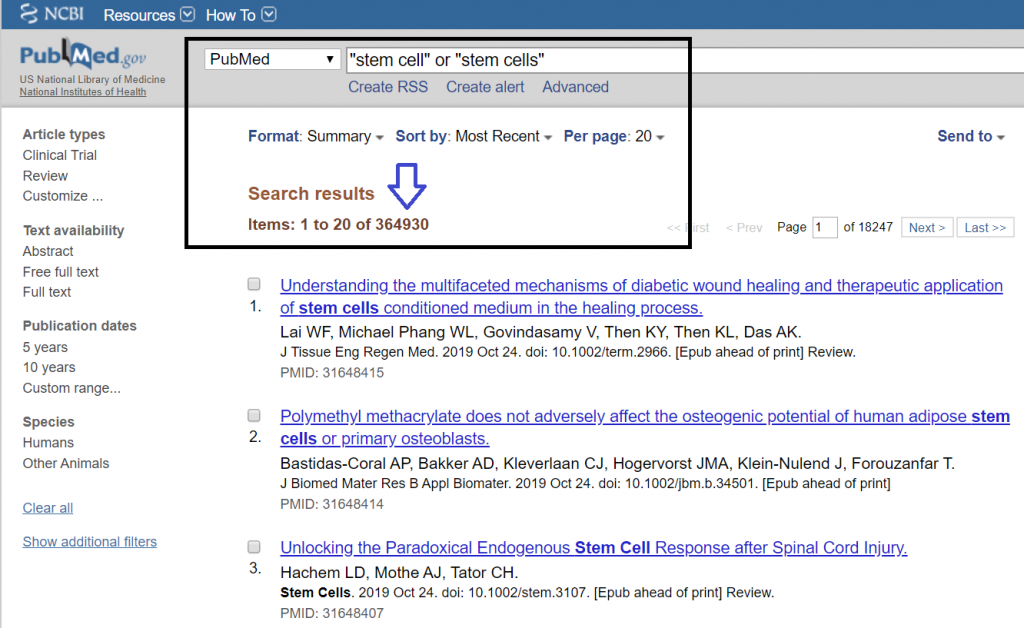
Stem cells have gained tremendous momentum over the last decade. As of November 2019, stem cell scientific publications have reached over 364,930 worldwide, with 25,000+ now being published per year.
For stem cell clinical trials, 7,400+ have been registered on ClinicalTrials.gov. Because this registry includes approximately 3/4ths of clinical trials worldwide, the total number of stem cell clinical trials worldwide is estimated to be approaching 10,000.

















