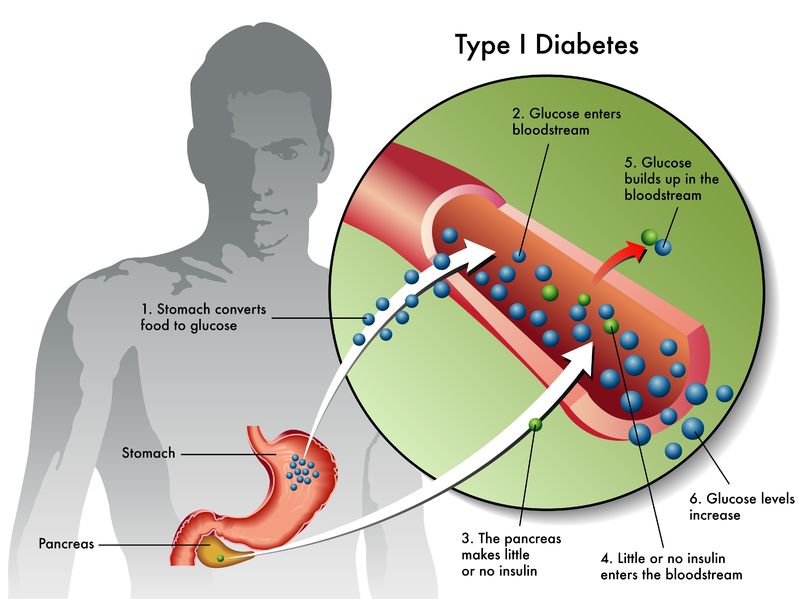
Australian stem cell company Cynata Therapeutics announced that it has filed an application to the UK Medicines and Healthcare products Regulatory Agency (MHRA) to commence a Phase I clinical trial for its lead product, CYP-001. CYP-001 is an iPSC-derived mesenchymal stem cell product that is anticipated to be the world’s first allogeneic iPSC product to enter a clinical trial.
Australian stem cell company Cynata Therapeutics announced that it has filed an application to the UK Medicines and Healthcare products Regulatory Agency (MHRA) to commence a Phase I clinical trial for its lead product, CYP-001. CYP-001 is an iPSC-derived mesenchymal stem cell product that is anticipated to be the world’s first allogeneic iPSC product to enter a clinical trial.
To learn more, read the full press release issued by Cynata Therapeutics, printed with permission from Cynata CEO, Dr. Macdonald. [Read more…]