In addition to commercial cell therapy CDMOs, there are other industry players that support the cell therapy industry, including academic and medical CDMOs. While these groups do contribute to the cell therapy CDMO market, their manufacturing capabilities are limited in scale and best suited to small, early-stage clinical trials. [Read more…]
Key Strategies for Positioning Cell Therapy CDMO Services – Size & Specificity
A contract development and manufacturing organization (CDMO), is a company that serves cell therapy companies on a contract basis. Common cell therapy CDMO services include cell therapy product development, manufacturing, clinical trial support, and commercial supply.
CDMOs allows cell therapy companies to outsource aspects of their business, which can support:
- Scalability
- Speed to market
- Adding technical expertise without overhead costs
- Cost efficiencies
Can New Manufacturing Platform Reduce “Sky High” Costs of Stem Cell Therapies?
The LA Times released a compelling article highlighting the mounting evidence that stem cell treatments will be some of the highest priced treatments within the medical marketplace. Titled “Sky-high price of new stem cell therapies is a growing concern,” the author Michael Hiltzik explores the often exorbitant costs associated with stem cell procedures.
In a powerful statement that summarizes the author’s position, he writes, “The evidence is already mounting that stem cell and other advanced biologic treatments will be among the most expensive therapies in the medical arsenal.” As examples of expensive cell therapy procedures, Hiltzik cites that Prochymal, a mesenchymal stem cell treatment approved in Canada, can costs as much as $200K, while Provenge, a cell-based vaccine for prostate cancer, can cost nearly $100K to extend a patient’s life by a few months. [Read more…]
Behind the Scenes at Cynata Therapeutics with Dr. Ross Macdonald
 Cynata Therapeutics Ltd (ASX: CYP) is a clinical stage regenerative medicine company specializing in stem cell therapeutics that has a proprietary technology for manufacturing induced pluripotent stem cell (iPSC) derived mesenchymal stem cells (MSCs).
Cynata Therapeutics Ltd (ASX: CYP) is a clinical stage regenerative medicine company specializing in stem cell therapeutics that has a proprietary technology for manufacturing induced pluripotent stem cell (iPSC) derived mesenchymal stem cells (MSCs).
I had the honor of interviewing Dr Ross Macdonald, CEO of Cynata Therapeutics. In this interview, we discuss Cynata’s technology platform, the company’s world first achievements, and its clinical trial underway for GvHD. We also explore the company’s strategic goals and the importance of its partnership with Fujifilm in Japan. [Read more…]
GenCure announces breakthrough first run of 80-liter bioreactor
Biomanufacturing CDMO among first to harvest adult stem cells at large scale
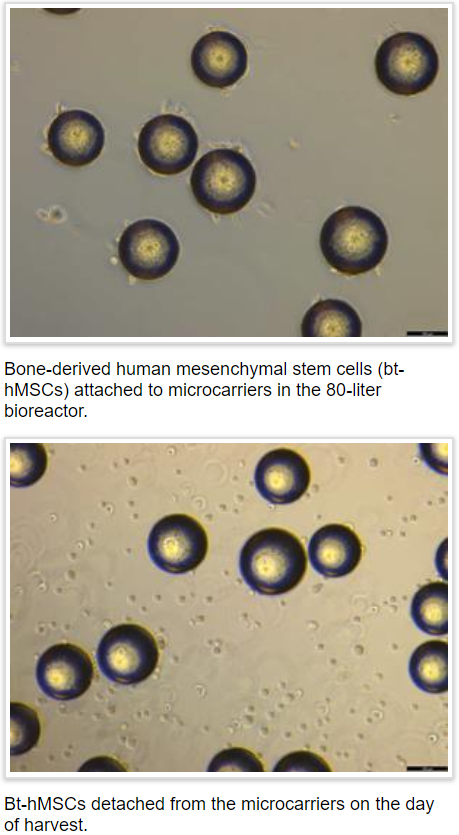
SAN ANTONIO, Texas – In a breakthrough for the regenerative medicine industry, GenCure announced today that its biomanufacturing services team has successfully completed production runs expanding and harvesting human mesenchymal stem cells (hMSCs) in an 80-liter bioreactor. GenCure, a subsidiary of BioBridge Global (BBG), announced a major expansion of its biomanufacturing facilities in February of this year. [Read more…]
- 1
- 2
- 3
- 4
- Next Page »





















