Cell therapy was recently described as “One of the most promising new fields of science and medicine,” by FDA Commissioner Scott Gottlieb. He went on to state that cell therapy and regenerative medicine “…hold significant promise for transformative and potentially curative treatments for some of humanity’s most troubling and intractable maladies.” In the FDA statement issued August 28th, Gottlieb also revealed that the U.S. regulator is looking improve upon its approval process for cell therapies. [Read more…]
Pluristem Granted Key U.S. Patent for Skeletal Muscle Regeneration – a Meaningful Asset Ahead of Upcoming Phase III Femoral Neck Fracture Study
- Patent relates to the use of Mesenchymal Stem Cells (MSCs) for muscle regeneration following muscle injury
- Pivotal Phase III study to take place in U.S. and Europe to support recovery from hip fracture
HAIFA, Israel, Nov. 22, 2017 (GLOBE NEWSWIRE) — Pluristem Therapeutics Inc. (NASDAQ:PSTI) (TASE:PSTI), a leading developer of placenta-based cell therapy products, announced today that the U.S. Patent and Trademark Office (USPTO) has issued a patent titled, “Skeletal muscle regeneration using mesenchymal system cells.” This key patent, which has already been granted in Europe, Hong Kong and Israel, addresses the use of MSCs for skeletal muscle regeneration used either directly after, or shortly after, post-surgical muscle injury.
 The Company received positive feedback from the U.S. Food and Drug Administration (FDA) and the European Medicine Agency (EMA) for the proposed study design and endpoints of its Phase III trial for the treatment of muscle recovery following arthroplasty for hip fracture. This planned study was recently awarded an $8.7 million grant by the Horizon 2020 program, the European Union’s largest research and innovation program. If successful, Pluristem plans to use the study results to achieve marketing approval in both the U.S. and Europe. [Read more…]
The Company received positive feedback from the U.S. Food and Drug Administration (FDA) and the European Medicine Agency (EMA) for the proposed study design and endpoints of its Phase III trial for the treatment of muscle recovery following arthroplasty for hip fracture. This planned study was recently awarded an $8.7 million grant by the Horizon 2020 program, the European Union’s largest research and innovation program. If successful, Pluristem plans to use the study results to achieve marketing approval in both the U.S. and Europe. [Read more…]
Pluristem Partners with Tel Aviv Sourasky Medical Center for Phase I/II Trial in Steroid-Refractory Chronic GvHD
- Tel Aviv Sourasky Medical Center will act as the sponsor of the study
- PLX-PAD cells have demonstrated efficacy in preclinical models of GvHD
HAIFA, Israel, Nov. 06, 2017 — Pluristem Therapeutics Inc. (NASDAQ:PSTI) (TASE:PSTI), a leading developer of placenta-based cell therapy products, today announced that it has signed an agreement with Tel Aviv Sourasky Medical Center (Ichilov Hospital) to conduct a Phase I/II trial in PLX-PAD cell therapy for the treatment of Steroid-Refractory Chronic Graft-versus-Host-Disease (GvHD).
 The trial will be an investigator initiated study. As such, Tel Aviv Sourasky Medical Center will support the study and will be responsible for its design and implementation. Dr. Ron Ram, Director of the Hematology Blood and Marrow Stem Cell Transplantation Unit for the Tel Aviv Sourasky Medical Center will act as principal investigator.
The trial will be an investigator initiated study. As such, Tel Aviv Sourasky Medical Center will support the study and will be responsible for its design and implementation. Dr. Ron Ram, Director of the Hematology Blood and Marrow Stem Cell Transplantation Unit for the Tel Aviv Sourasky Medical Center will act as principal investigator.
GvHD is a potentially lethal complication of hematopoietic cell transplantation (HCT) from a donor. When a patient receives a donor’s stem cells, the transplanted cells identify the patient’s body as foreign and attack it. The chronic form of GvHD occurs at least 100 days following the transplantation. The GvHD market is predicted to exceed $500 million by 2023. [Read more…]
FDA Grants Pluristem Orphan Drug Designation for Its PLX-R18 Cell Therapy as Treatment for Acute Radiation Syndrome
Pluristem’s ARS program is developed and funded by the U.S. National Institutes of Health and the U.S. Department of Defense, and is in preparation for a pivotal study
HAIFA, Israel, Oct. 19, 2017 — Pluristem Therapeutics Inc. (NASDAQ:PSTI) (TASE:PSTI), a leading developer of placenta-based cell therapy products, announced today that the U.S. Food and Drug Administration (FDA) has granted the company an orphan drug designation for its PLX-R18 cell therapy for the prevention and treatment of acute radiation syndrome (ARS). [Read more…]
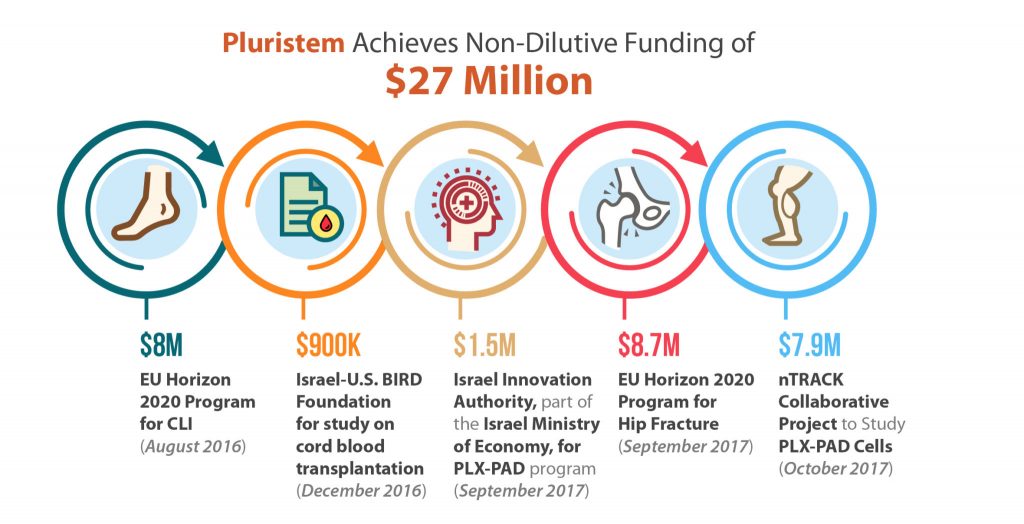
Pluristem Lands $27 Million in Non-Dilutive Funding for its Advanced-Stage Cell Therapies
Non-dilutive funding, in substantial amounts, is something nearly all biotechnology companies want, and few get. Although small grants from various government agencies like the NIH are helpful, and are available to a larger number of biotechs, Pluristem Therapeutics (Nasdaq:PSTI) has accomplished something quite unique in the last year as the recipient, of three awards totaling nearly $25 million from the European Union’s Horizon 2020 program as well as $2.4 million in smaller grants from Israel. A majority of these funds are aimed at bringing Pluristem’s Phase III assets to market and to better predict outcomes of cell therapy treatments.
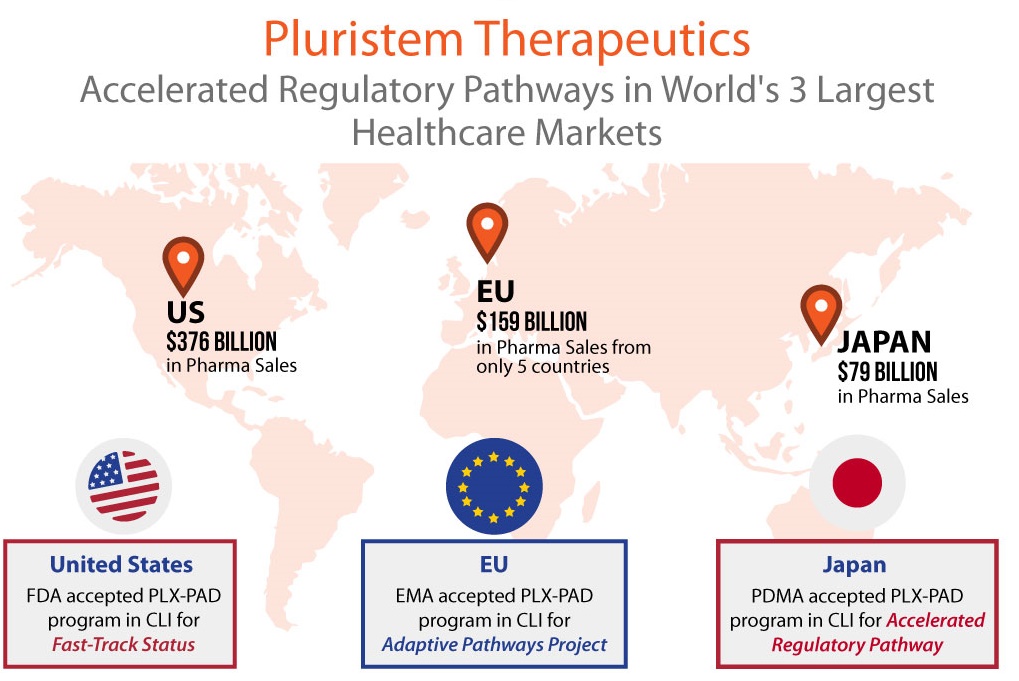
Pluristem’s lead asset, its PLX-PAD cell therapy, is currently in a multinational pivotal Phase III study for the treatment of critical limb ischemia (CLI) in 250 patients with CLI Rutherford Category 5 who are unsuitable for revascularization. These patients have very few remaining treatments options and will most likely be headed for amputation. Cell therapy may be the answer. The U.S. FDA has given Pluristem Fast Track Designation in this indication, while the European Medicines Agency (EMA) has selected the PLX-PAD program in CLI for its Adaptive Pathways project, the goal of which is to shorten time to market for innovative medicines. Europe gave the PLX-PAD program in CLI one more boost with the award of $8 million last August from the EU’s Horizon 2020 program, Europe’s largest research and innovation program. Clearly, the Europeans are eager for a novel cell therapy to treat CLI and they are banking on Pluristem to bring it to market quickly. [Read more…]