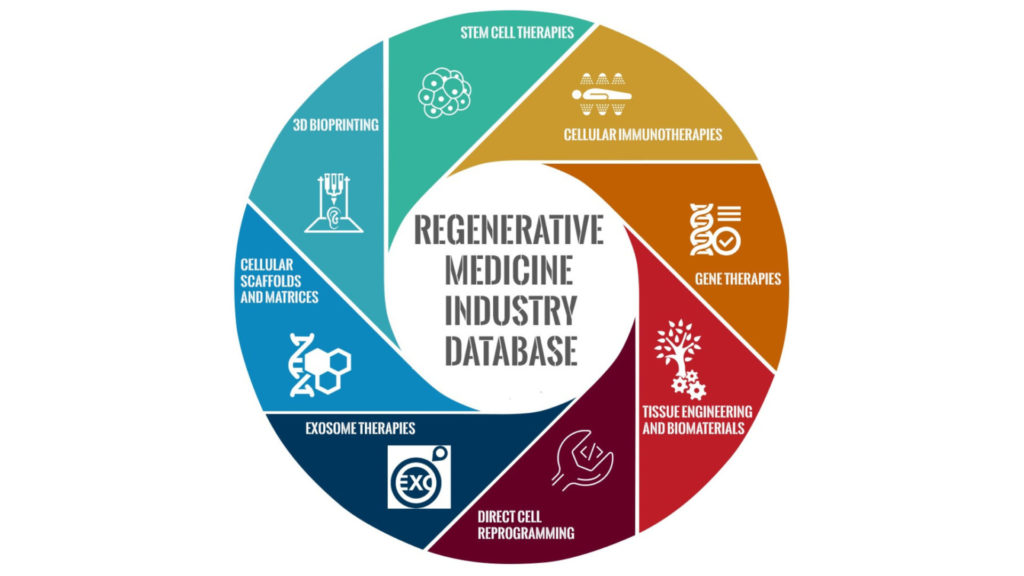
The regenerative medicine industry is a high-value, fast-growth market. With two CAR-T approvals, as well as the first gene therapy and digital pill approvals in the United States, we have had a lot of FDA “firsts.” The market gathered momentum when the Swiss pharmaceutical giant Novartis made history as the first company to win FDA approval for a CAR-T cell therapy in the U.S. in August 2017 (Kymriah). In October 2017, Kite Pharma became the second company to get FDA approval of a CAR-T cell therapy (Yescarta), further “snowballing” the effect. [Read more…]
How iPSCs are Becoming an Emerging Area of Stem Cell Research
Since the discovery of induced pluripotent stem cells (iPSCs) in 2006, a large and diverse market for iPSC research tools has emerged. Included below is a ten-year history of iPSC grants (source: RePORT.NIH.gov), clinical trials (source: ClinicalTrials.gov), and scientific publications (source: PubMed.gov).
Both graphs below reveal the iPSC market is expanding. [Read more…]
2017 Marks the 1st Year That Regenerative Medicine Disrupted Healthcare
The regenerative medicine industry has been rapidly expanding over the past few years, making it a high-value, fast-growth market. Key drivers for the market include high rates of clinical trials, accelerated pathways for product approvals, new technologies to support cell and gene therapy manufacturing, and the potential for cell therapies to revolutionize healthcare.
The regenerative medicine market gained major momentum when the Swiss pharmaceutical giant Novartis made history as the first company to win FDA approval for a CAR-T cell therapy in the U.S. in August 2017 (Kymriah). In October 2017, Kite Pharma became the second company to get FDA approval of a CAR-T cell therapy (Yescarta).
With swelling momentum to support the CAR-T technology, there are now close to 40 companies developing redirected T cells or NK cells for therapeutic use. [Read more…]
RoosterBio Launches New cGMP Stem Cell Product Portfolio
RoosterBio Launches New cGMP Stem Cell Product Portfolio, Maintaining Unwavering Commitment to Driving the Explosive Regenerative Medicine Industry
Next Generation Products are Harbinger of Paradigm Shift in the Adult Stem Cell Industry
 Frederick, MD, USA. September 14, 2017 – RoosterBio Inc, today, proudly announces the launch of its CliniControl™ (CC) product portfolio, clinically-relevant starting and ancillary materials that generate unprecedented efficiencies in adult stem cell manufacturing, accelerating clinical translation of human Mesenchymal Stem/Stromal Cell (hMSC)-based therapies. The CliniControl product portfolio will radically simplify major steps in product development and clinical translation, improving the likelihood of commercial success for RoosterBio customers.
Frederick, MD, USA. September 14, 2017 – RoosterBio Inc, today, proudly announces the launch of its CliniControl™ (CC) product portfolio, clinically-relevant starting and ancillary materials that generate unprecedented efficiencies in adult stem cell manufacturing, accelerating clinical translation of human Mesenchymal Stem/Stromal Cell (hMSC)-based therapies. The CliniControl product portfolio will radically simplify major steps in product development and clinical translation, improving the likelihood of commercial success for RoosterBio customers.
The first in the CliniControl line of products, RoosterNourish™-MSC-CC bioprocess medium sets the new standard of industry-leading performance. RoosterNourish-MSC-CC is a first-in-class, standardized, xeno-free (XF) bioprocess medium engineered to support not only 2D (plastic adherent), but also 3D suspension culture of hMSCs in allogeneic and autologous therapeutic manufacturing. Formulated specifically for clinical research process and product developers, RoosterNourish-MSC-CC yields superior media productivity, leading to drastically reduced cost of goods as compared to other hMSC culture media. The highly productive RoosterNourish medium is designed for batch 2D and fed-batch 3D cultures and seamlessly translates between culture platforms enabling scalable hMSC culture, delivering robust, reproducible and standardized results at a commercially-viable cost of goods. [Read more…]
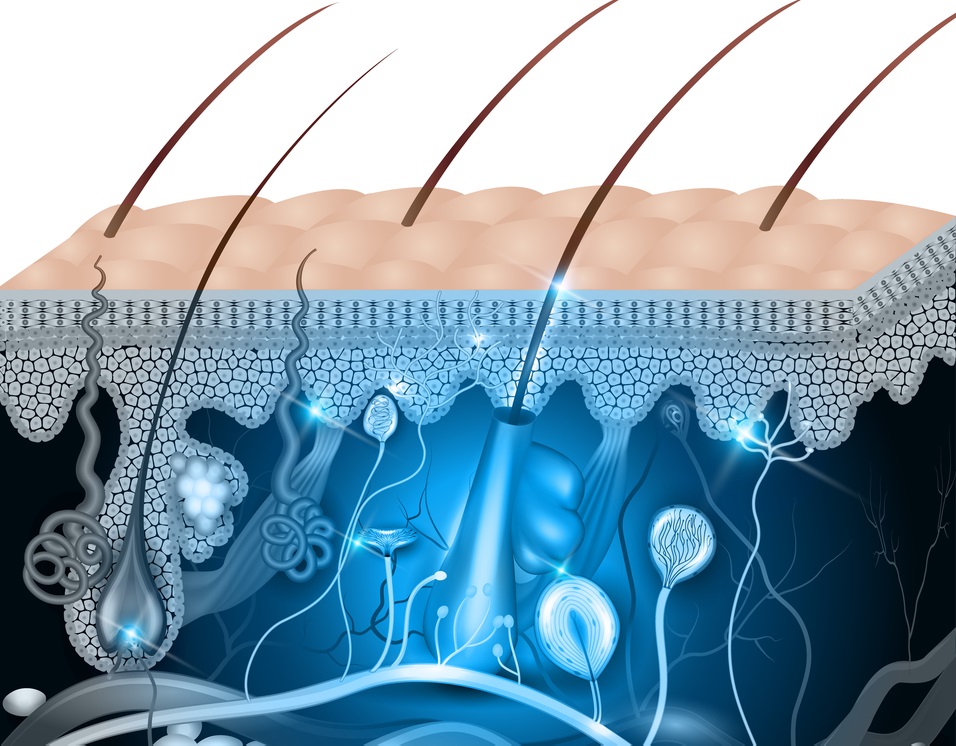
PolarityTE(TM) Regenerates Full-Thickness Hair-Bearing Skin in Burns & Wounds Using Revolutionary Platform Technology
First ever known successful regeneration of full-thickness skin and hair; Company poised to initiate human trial in the third quarter of 2017; Management to host conference call Thursday, June 8th at 4:30pm ET
 SALT LAKE CITY, UT — 06/08/17 — PolarityTE™, Inc. (NASDAQ: COOL) today announced pre-clinical results demonstrating that the Company’s lead product, SkinTE™, regenerated full-thickness, organized skin and hair follicles in third degree burn wounds. The findings represent the first known successful regeneration of skin and hair in full-thickness swine wound models, the standard animal model for human skin. The Company expects to initiate a human clinical trial evaluating the autologous homologous SkinTE™ construct in the third quarter of 2017. [Read more…]
SALT LAKE CITY, UT — 06/08/17 — PolarityTE™, Inc. (NASDAQ: COOL) today announced pre-clinical results demonstrating that the Company’s lead product, SkinTE™, regenerated full-thickness, organized skin and hair follicles in third degree burn wounds. The findings represent the first known successful regeneration of skin and hair in full-thickness swine wound models, the standard animal model for human skin. The Company expects to initiate a human clinical trial evaluating the autologous homologous SkinTE™ construct in the third quarter of 2017. [Read more…]
- « Previous Page
- 1
- 2
- 3
- 4
- …
- 13
- Next Page »