Human mesenchymal stem cells (hMSCs) are multipotent adult stem cells present in a variety of tissue niches within the human body. As multipotent cells, they can differentiate into a range of cell types, including osteoblasts, chondrocytes, myocytes, and adipocytes. In addition to secreting factors that can stimulate tissue repair, MSCs can substantially alter their microenvironment, exerting effects that are both anti-inflammatory and anti-fibrotic.
Advantages of Human MSCs
hMSCs have a unique capacity for use in therapeutic applications because they are immuno-privileged, can be readily sourced from a variety of tissues, and have the ability to migrate to tumors and wounds in vivo. Additionally, they avoid the ethical issues that surround the embryonic stem cell research. They are well-suited for use in the exponentially growing area of 3D printing, due to their unique capacity to form human structural tissues.
Because of these advantageous traits, MSCs have been cited in more than 47,000 scientific publications released to date. There are also close to one-thousand MSC trials underway worldwide, with East Asia, Europe, and the United States representing hot-spots for a clinical trial activity.
Clearly, hMSCs have become desirable tools for use in tissue engineering and cell-based therapy applications.
hMSC Culture Medium as An Ancillary Material for Cell-based Therapies
In most clinical applications, hMSCs are expanded in culture before use. Therefore, the quality of the culture medium and its performance are crucial in regard to therapeutic use, because hMSC properties can be significantly affected by medium components and culture conditions.
Most of the common culture media for growth and expansion of hMSCs, as well as auxiliary solutions that facilitate attachment, dissociation, and cryopreservation, are supplemented with serum or other xenogeneic compounds.
Therefore, a defined serum-free (SF), xeno-free (XF) culture system optimized for hMSC isolation and expansion would greatly facilitate the development of a robust, clinically acceptable culture process for generating quality-assured cells.
BI’s NutriStem® MSC Xeno-Free Culture Medium
Headquartered in Israel, Biological Industries is a multinational life science supplier with more than 30 years of experience in cell culture and MSC media development and manufacturing.
To meet the challenge of translating hMSC research into therapeutic applications, Biological Industries offers a novel xeno-free (XF) culture system comprising MSC NutriStem® XF culture medium and all of the required auxiliary solutions to support the attachment, dissociation, and cryopreservation of hMSC.
The system has been evaluated for the initial isolation of hMSC from various sources, as well as for long-term culturing under SF, XF culture conditions suited for clinical applications.
The use of the NutriStem system maintains the hMSC features that include the following:
- typical fibroblast-like cell morphology
- phenotypic surface market profile
- differentiation capacity
- self-renewal
- genetic stability
Additionally, research shows that Biological Industries’ MSC NutriStem XF culture system efficiently supports the initial isolation and optimal expansion of hMSC from various sources, as demonstrated by Dr. Genser-Nir, et al., in the article, “A Xeno-Free Culture System for hMSC from Various Sources Suitable for Initial Isolation and Expansion Toward Clinical Applications”
Drug Master File (DMF) for MSC NutriStem®
In 2015, Biological Industries (BI) submitted a Drug Master File (DMF) to the U.S. Food and Drug Administration (FDA) for purposes of moving its MSC NutriStem XF Basal and Supplement toward being certified for clinical applications.
A DMF is prepared by a pharmaceutical manufacturer and submitted to the regulatory authority in markets where the product is sold. While there is no regulatory requirement for DMFs, the document provides regulators with confidential, detailed information about facilities, processes, or articles used in the manufacturing, processing, packaging, and storing of the products.
For regulatory purposes, MSC NutriStem XF is classified as an “ancillary material”, which means it is a material used in the process, but is not part of, the final product. In accordance with the four-tier USP Risk classification system, the medium is characterized as low-risk if produced in compliance with GMP’s.
Biological Industries’ submission of the DMF indicates the company’s ongoing commitment to providing cell culture clients with unparalleled innovation, quality guarantees, and regulatory assurance. In addition to guaranteeing lot-to-lot repeatability, all NutriStem clients receive a certificate of analysis and DMF cross-reference. The product is also produced to superior quality standards, extensively quality control (QC) tested, and functionally characterized for performance in relevant cell models.
These high production standards are unparalleled within the life science marketplace.
Give Your Cells a Competitive Edge
As mentioned, a successful culture of stem cells requires optimized protocols that incorporate high quality, xeno-free reagents. While mesenchymal stem cell (MSC) research represents a rapidly progressing area of medicine, clinical applications remain limited by xeno contamination during the in vitro derivation and propagation phases.
Bridging the gap between research and therapeutic applications requires the design and implementation of qualified protocols and operating processes. Making xeno-free and animal component-free media is an essential component of translating hMSC therapies into human subjects.
For this reason, BI developed its MSC NutriStem XF medium as a defined, serum-free, xeno-free culture medium to promote optimal growth and expansion of human mesenchymal stem/stromal cells (hMSC) derived from a variety of sources, including but not limited to the following:
- bone marrow (BM-MSC)
- adipose tissue (AT-MSC)
- umbilical cord matrix (UC-MSC)
- dental pulp (DP-MSC)
hMSCs cultured in MSC NutriStem XF medium maintain proper fibroblast-like cell morphology, tri-lineage differentiation potential, and demonstrate normal hMSC marker profiles and karyotypic stability over long-term culture. They also show superior proliferation and self-renewal potential in comparison to serum-containing media and other commercial serum-free media.
NutriStem MSC XF medium is designed for optimal growth and expansion of hMSCs derived from a variety of sources, including bone marrow (BM-hMSC), adipose tissue (AT-hMSC), Wharton’s jelly (WJ-hMSC), placental tissue (PT-MSC), and umbilical cord matrix (UC-hMSC).
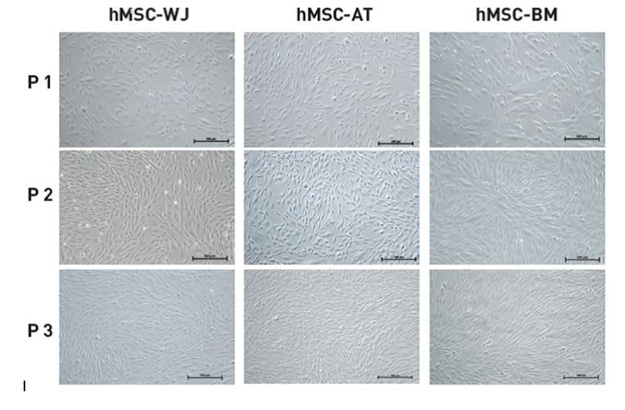
Demonstration of hMSCs in 3 Passages of the XF Culture System
As demonstrated below, hMSCs derived from WJ, AT, and BM are cultured for 3 passages in the XF culture system (MSC NutriStem XF, MSC attachment solution, and MSC dissociation solution). Representative images were taken on Day 3 of culture (x100). MSC NutriStem XF promotes proliferation of hMSC from a variety of sources while maintaining their fibroblast-like morphology.
Similarly, Dr. Weiss, et al., compared three commercially prepared serum-free (SF) media and a 2% serum containing growth medium (SGM) to determine expansion, phenotypic stability, and multipotency. He concludes that Biological Industries’ MSC NutriStem XF and substrate provide optimal umbilical cord-derived hMSCs expansion as compared to other commercially available serum-free (SF) formulations, including those produced by Stemcell Technologies and Invitrogen. Full findings can be found in the article “Identification of Optimal Conditions for Generating MSCs for Preclinical Testing: Comparison of Three Commercial Serum-Free Media and Low-Serum Growth Medium”.
Traits of MSC NutriStem® XF
In summary, the following traits make BI’s MSC NutriStem XF ideal for isolation and growth of hMSCs.
- an ancillary material for cell therapy and has been used for clinical applications
- produced in a cGMP compliant facility
- accompanied by a Drug Master File (DMF)
- suitable for hMSC from various tissues
- xeno-free
As hMSC clinical trials continue to multiply in number, it is clear that this new serum-free (SF), xeno-free (XF) culture system optimized for hMSC isolation and expansion will greatly facilitate the translation of hMSC research into clinically relevant therapies.
About Biological Industries Ltd.

Biological Industries (BI) develops and manufactures life science products for universities, government research, healthcare institutions, and the biopharmaceutical industry. Supported by in-house R&D, 30 years of experience, and academic collaborations, BI has introduced a series of serum-free media and many other products for animal cell culture and molecular biology. It developed NutriStem® — a serum-free, xeno-free, gold standard, stem cell culture media for optimal growth and expansion of pluripotent stem cells and mesenchymal stem cells. Learn more at http://www.bioind.com/.




















Tell Us What You Think!