|
|
Direct cell reprogramming is the in vitro (laboratory) or in vivo (in the body) reprogramming of somatic cells into other cell types without the need for an intermediate pluripotent state. Another term for direct cell reprogramming is transdifferentiation, which is the direct conversion of one differentiated cell type into another.
There has been tremendous activity over the last decade in the development of stem cells therapeutics, which have relied on stem cell differentiation protocols, as well as techniques for direct cell reprogramming. Today, there are over 7,149+ stem cell trials registered on ClinicalTrials.gov and stem cell publications have increased dramatically in recent years, recently surpassing 500,000+ on PubMed.gov.
However, for all their promise, stem cells are yet to deliver on this therapeutic potential, and this failure appears due to various technical challenges that still need to be overcome.
In this article:
- Origins of Direct Reprogramming
- Totipotent, Pluripotent, and Multipotent
- iPS Cells: Therapeutic Potential Limited
- Direct Reprogramming of Autologous Multipotent Stem Cells
- Direct Cell Reprogramming: In vitro vs. in vivo
- Autologous Reprogrammed Cells Better than Allogeneic Cells
- Future of Direct Cell Reprogramming: Mogrify and Asgard Therapeutics
The Origins of Direct Reprogramming
To explain direct reprogramming, we need to go back to the beginning.
The cell is the basic building block of all living organisms and the human body is composed of trillions of cells. They provide structure for the body, take in nutrients from food, convert those nutrients into energy, and carry out specialized functions. These functionalities are determined by the genes that are expressed in that particular cell.
All cells in the body contain the same genes (~22,000), but only certain ones are “expressed” in each particular cell, making the proteins that determine the functions of that cell.
A group of different cells working together to fulfill a function constitutes a tissue (e.g., neurons-astrocytes-oligodendrocytes working synergistically constitute neuronal tissue). A group of tissues working together to fulfill a bodily function constitutes an organ (e.g., the brain or the spinal cord). Finally, a concerted group of organs are re-grouped in a system (e.g. the brain and spinal cord constitute the Central Nervous System).
If cells make tissues, organs and systems, then what makes cells?
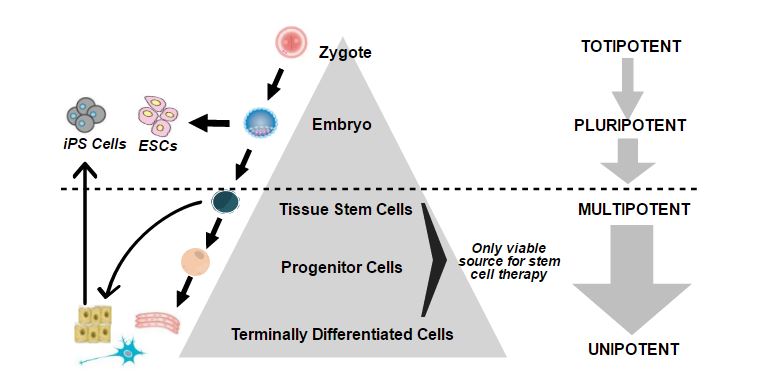
Stages of Development: Totipotent, Pluripotent, and Multipotent cells
At the beginning of human life when the sperm fertilizes the egg, totipotent cells are born of the zygote. They possess the ability to become anything (placenta and embryo) and are present in such form up to the 16-cell stage (i.e., within the first couple of cell divisions after fertilization, up to 4 days post fertilization). Following this stage of totipotency, as the number of cells increases, pluripotent cells emerge.
Pluripotent cells retain the ability to make any cell of the embryo (embryonic stem cells are pluripotent), but lose the ability to become placenta.
After 3 weeks post fertilization, pluripotent cells begin to organize themselves into 3 germ layers (Ectoderm, Mesoderm, and Endoderm) and progressively differentiate towards a more defined state. Pluripotent stem cells have attracted significant attention since Yamanaka was able to reprogram “mature” fibroblasts (skin cells) to pluripotent stem cells (induced pluripotent stem cells or iPS cells).[1] This pioneering work led Yamanaka to receive the Nobel Prize for Medicine in 2012.
However, iPS cells were abandoned by most scientific investigators as therapeutic candidates themselves, given their poor safety profile. (They go back to a pluripotent state, and in addition to the desired tissue, also form unwanted teratomas, which are rapidly growing tumors containing cells of all three germ layers). In fact, significant efforts have gone into attempting to differentiate these iPS cells into multipotent stem cells that are safe and potent therapeutic cell candidates.
Multipotent stem cells are specialized stem cells differentiated from one of the three germ layers, but restricted within a specific lineage. Each type of multipotent stem cell makes the specialized somatic cells of its lineage: for example, a neural stem cell is a multipotent stem cell that originates in the ectodermal germ layer and is the only multipotent stem cell that has the ability to make neurons, astrocytes and oligodendrocytes (and thus, neuronal tissue), and does not have the ability to form any other tissue.

iPS Cells: Therapeutic Potential Limited by Safety, Potency, and Cost
iPS cells promised to be a boon for regenerative medicine. Researchers could take a person’s skin, blood, or other cells, reprogram them into iPS cells, and then use those to grow liver cells, neurons, or whatever was needed to treat a disease. This personalized therapy would get around the risk of immune rejection, and sidestep the ethical concerns of using cells derived from embryos and fetuses.
Unfortunately, the reality has been very different. Producing autologous iPS cell derived multipotent stem cells at a scale that can be used for clinical purposes has proved to be very difficult, time consuming, and costly. The only clinical trial using autologous iPS cell multipotent stem cells was halted in 2015 after only one person had received a treatment. As the lab prepared to treat the second trial participant, Yamanaka’s team identified two small genetic changes in both the patient’s iPS cells and the retinal pigment epithelium (RPE) cells derived from them.
There was no evidence that either mutation was associated with tumor formation, yet “to be on the safe side,” the trial was put on hold. In June 2016, the trial resumed, but was switched to allogeneic, rather than autologous cells, presumably for purposes of cost and time efficiency, as well as safety. Thus, 10 years on from the discovery of iPS cells, iPS cells are no longer viewed as the key source of therapeutic cell candidates. They have become an important tool for modelling and investigating human diseases, as well as for screening drugs.
iPS cells have become a lab workhorse — providing an unlimited supply of once-inaccessible human tissues with specific genetic mutations for research.
Direct Reprogramming of Autologous Multipotent Stem Cells
Today, autologous multipotent stem cell therapy is regarded by many as the “Holy Grail” for regenerating and replacing damaged, lost, or aged cells in organs. Bone marrow and fat (adipose) stem cells are effective therapeutic multipotent stem cells for replacing bone, cartilage and fat cells. However, they do not have the ability to replace cells in other organs, where cells are derived from other lineages.
Direct cell reprogramming is uniquely designed to allow a patient’s cell to “jump” directly to the specialized multipotent stem cell of choice, thus tailor-making autologous multipotent stem cells needed to repair a damaged or diseased organ.
Instead of making pluripotent stem cells from a patient’s cells and differentiating them to multipotent stem cells over an extended period of time, direct cell reprogramming produces directly the specialized multipotent stem cell of choice, solving the safety, high cost, and time constraints associated with iPS cells.

In short, direct cell reprogramming is the reprogramming of somatic cells into other cell types without the need for an intermediate pluripotent state.
Another term for direct cell reprogramming is transdifferentiation, which is the direct conversion of one differentiated cell type into another. The process does not require an intermediate pluripotent state or progenitor cell to be created.
Direct Cell Reprogramming | In vitro vs. in vivo
The main approach for direct cell reprogramming is based on in vitro reprogramming of harvested somatic cells from an accessible source (blood, skin, or bone marrow) to the specialized multipotent stem cell type of choice, according to the indication or disease (i.e. a neural stem cell for CNS conditions, a cardiac stem cell for heart disease, etc.). This approach relies on using transcription factors and chromatin remodeling agents to permanently alter the expression patterns of the cells, locking them into their new multipotent stem cell state of choice.
The key to ultimate clinical success with this approach involves selecting the most relevant cell subtype(s) for reprogramming, refraining from using genetic manipulation or viral vectors for changing the gene expression pattern, and achieving stable reprogramming.
A second approach being investigated (chemically induced direct reprogramming) relies on using small molecules in vivo (drugs directly in patients) to induce cell-lineage reprogramming. Such a strategy could be cost-effective and might sound attractive. However, specific targeting of systemically administered small molecule drugs to specific cells/tissues (without affecting other cells or parts of the body) is still many years away.
Even after decades of research by pharmaceutical firms, targeted drug delivery remains a key problem for the industry. Hence, in vivo this approach would raise significant safety concerns, because the drugs could result in widespread effects (e.g. turning many or most of the cells within the body into neurons).
Directly reprogrammed multipotent stem cells are destined to be lineage-restricted stem cells that regenerate and grow defined tissues of their specific lineage.
They provide “new seeds” that can actually re-grow the targeted damaged tissue or organ. (This unique feature is sometimes confused with the pluripotent stem cells that do not possess this feature, but instead can differentiate into some unwanted cell types too). This restricted ability also means that the right type of stem cell can and should be used in the particular tissue or organ to be treated. For example, hematopoietic or mesenchymal stem cells have limited regenerative and no cell replacement abilities if implanted into the CNS, whereas neural stem cells are the cells that have been evolved for this specific purpose.
General lack of awareness surrounding this feature has resulted in confusion, whereby for example, mesenchymal stem cells (MSCs) have been implanted into numerous unrelated tissues (e.g. the brain), and this has resulted in, as expected, limited beneficial effects. This approach can also be harmful, as one patient that received these MSCs injected into her eye actually started growing bone inside her eye.
Autologous Reprogrammed Cells Better than Allogeneic Cells
For any stem cell to correctly, efficiently, and permanently replace cells in a patient’s tissue, the stem cell has to be of the correct lineage and autologous (the patient’s own cell in the right organ or tissue), and adequately “young” to be able to do the job. So far, this has not happened within the stem cell field. The only exception to this is hematopoietic stem cell grafts (bone marrow transplants) that have been performed since the 1960s to support the cure of blood diseases, such as leukemia, a rare example of where allogeneic stem cells can be used.
The procedure is more akin to a full organ transplant than to cell therapy, as the entire hematopoietic system is replaced after irradiation (killing off) of the patient’s old hematopoietic system. In the process, the white cells that would normally reject an allogeneic graft are killed off too.
Of course, this irradiation of the “native” tissue cannot be done for tissue such as the brain. It is important to note that even with closely matched hematopoietic stem cell (HSC) transplants, the success rate of these procedures is often less than 20%, even after more than 50 years of clinical refinement. As a comparison, autologous hematopoietic stem cell transplants (from cord blood) have had no graft failures. Still, this partial exception of hematopoietic stem cell transplants has unfortunately caused confusion about the abilities of other donor stem cells to avoid rejection.
For example, there is a perception that other types of donor stem cells will adequately graft into a patient if the patient is on immunosuppressants or if the particular type of somatic stem cells has a very low immune signature (as is, for example, the case with mesenchymal stem cells). The reality is that although these strategies allow donor stem cells to survive in the patient’s body for a period of time without being fully detected by the patient’s immune system, they are still ultimately killed off. In addition, any immunosuppressive regimen can cause serious side effects in patients.
As more is understood about the mechanisms of direct cell reprogramming, their specialized manufacturing, and the importance of autologous lineage-restricted stem cells, these unique multipotent stem cells have the potential to drive regenerative medicine into the clinic around the world and compliment small molecule drugs, proteins, oligonucleotides and medical devices, in tomorrow’s medical portfolio. This is good news for patients afflicted with some of the most challenging diseases and injuries, as well as for society at large, which is crumbling under the cost of chronic care management.
The Future of Direct Cell Reprogramming
What is the future of direct cell reprogramming? Most likely, it will offer an alternative therapeutic approach to some types of cell therapies, such as stem cell therapies and immunotherapies.
For example, a fascinating company that is working in this area is Mogrify, a private biotechnology company that has raised over $40M USD to advance its direct cell reprogramming platform. According to the company, Mogrify hopes to “Identify the optimal combination of transcription factors (in vitro) or small molecules (in vivo), needed to convert any mature cell type into any other mature cell type without going through a pluripotent stem cell- or even a progenitor cell-state.”
Mogrify also has a collaborative research agreement with Astellas Pharma, a global leader in regenerative medicine, to develop in vivo approaches to treating sensorineural hearing loss.
Asgard Therapeutics is an exciting a Sweden-based biotech company that specializes in vivo direct cell reprogramming immunotherapies for the treatment of cancer. In March 2024, it secured €30M in Series A financing that will support the development of its gene therapy, AT-108. AT-108 directly reprograms cancer cells inside the patient’s body to become Type 1 Dendritic Cells (cDC1s), creating a personalized and potent anti-tumor immune response.
Although their website no longer appears to be working, Fortuna Fixa was another a clinical biotech company that had specialized in cell reprogramming related to Parkinson’s disease and spinal cord injuries. The company produced two remarkable pieces of technology: drNPC, an autologous directly reprogrammed neural cell, and RMx™, a regeneration matrix. Fortuna Fixa’s LinkedIn page is still active and can be viewed here.
The challenges facing cell reprogramming are substantial, but a future in which a person own cells can be reprogrammed in vivo into therapeutic cells is an incredible future to consider.
If you found this blog valuable, subscribe to BioInformant’s stem cell industry updates.
Do you have questions about direct reprogramming? Ask them in the comments below.
|1. Cell. 2006 Aug 25;126(4):663-76. Epub 2006 Aug 10. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Takahashi K, Yamanaka S.

Cade, Great post! You said “Today, autologous multipotent stem cell therapy is regarded by many as the “Holy Grail” for regenerating and replacing damaged, lost, or aged cells in organs. Bone marrow and fat (adipose) stem cells are effective therapeutic multipotent stem cells for replacing bone, cartilage and fat cells. However, they do not have the ability to replace cells in other organs, where cells are derived from other lineages…” But, isn’t it true that researchers are telling us that these MSCs primarily *orchestrate* the regeneration of tissue, but do not incorporate into the tissue or organ as their primary mode of action–or am I wrong? I seem to remember some research presented at WSCS this year that only 5% of the MSCs in some autologous stem cell studies incorporated into the regenerated tissue. Thoughts? Sincerely, Dr. Bauman
Many thanks, Dr. Bauman. To regenerate damaged or diseased tissue, the therapeutic stem cells have to be of the same lineage as the tissue they aim to regenerate/repair. Mesenchymal stem cells will provide fertiliser (growth factors etc.) that will help repair tissue of a different lineage, but they will not provide the seeds (new cells) to regenerate tissue. In some cases, that may be enough. But for optimal regeneration, you need autologous multipotent stem cells or progenitor cells of the right lineage/tissue type.
Cade – This is a very insightful, informative, and thought-provoking article. It’s great that you crossected the related science with patient implications and market forces. In speaking with physicians, I am seeing that there is not strong consensus with regards to the best approach to do stem cell therapies. But, you pointing to the autologous approach as the “Gold Standard” or “Holy Grail” is consistent with what these physicians have communicated to me. I’ve worked in many fields, including telecommunications, healthcare diagnostics, and the U.S. military. With every field there are standards or “best practices” for each procedure, operation, or manufacturing process. Given this, it seems to me that a universally accepted standard for stem cell treatments is needed. Perhaps there is a need for a world conference on this matter for the medical/scientific community to come together and define, document, and certify the best approaches, techniques, and procedures for stem cell treatments. Perhaps this is in the works or has happened in some form, but clearly there is a need for further discussion, collaboration, and standard setting. Thanks for this valuable update!
Cade – Thank you for this article. It is well researched and well written. You mentioned that “autologous multipotent stem cell therapy is the Holy Grail…” and I completely agree with this statement. The research is showing amazing success results in patients using autologous multipotent Mesenchymal stem cells. You also mentioned bone marrow and adipose tissue as sources for these cells however you missed one very important source which is teeth. The reason this is important to your article is that the functional ability of MSC’s derived from different places in the body are not identical and the cells derived from children’s deciduous teeth have been proven superior to the cells obtained from other sources. The success of regenerative medicine relies primarily on the functionality and health of the starting material so better starting material is very important. It is also important to note that every single person loses their baby teeth naturally thereby providing an accessible supply without an invasive harvest procedure.
You also said; “Bone marrow and fat (adipose) stem cells are effective therapeutic multipotent stem cells for replacing bone, cartilage and fat cells. However, they do not have the ability to replace cells in other organs, where cells are derived from other lineages.” It is well established that MSC’s are able to direct the repair of non-related tissue and also to be able to differentiate into some tissues of non-mesenchymal lineage. Two examples of this are their ability to form different types of nerve cells and to form insulin secreting cells, both of which have been used in human patients.
There is a tremendous amount of work that needs to be done before clinicians know the best methods of utilizing this material to improve patient health. However, there are some amazing success stories out there already proving that MSC’s can be safely used in human patients and that they just may be that “Holy Grail” of medicine we are searching for.
Great comment, Dr. Byrom. Dental pulp is definitely a valuable source of MSCs, in addition to bone marrow and adipose tissue. Thank you for highlighting the advantages of MSCs from deciduous teeth. There are some publications showing that MSCs (and some other cells) can transdifferentiate into insulin-secreting cells and into potential neurons. However, they do not possess all the characteristics of beta cells and neurons, are generally maintained by forced expression of factors in the media that they are grown in, and will revert back to MSCs (or their bone, cartilage and/or adipose cells) when they are placed back into MSC media (or media for bone, cartilage and/or adipose cells). The cells are, thus, not true beta cells or neurons. Their efficacy and ability to maintain their state long-term has also not been shown in well-controlled human trials. Remember also that bone marrow contains, for example, a small number of neural crest cells and other cells types that were confused with MSCs in early publications.
Of course, it is possible to turn MSCs (and other cell types) into beta cells or neural stem cells and neurons via the new technology of direct reprogramming, which is the subject matter of some recent publications that are very interesting.
There are many interesting opportunities for stem cell therapeutics, positioning us at an exciting point in history.
Dr. Bauman, you are right, if well “Bone marrow and fat (adipose) stem cells are effective therapeutic multipotent stem cells for replacing bone, cartilage and fat cells. However, they do not have the ability to replace functional cells in other organs, but stem cell can replace the organ not functional (mechenquymal) tissue where cells are derived from other lineages”, the success effect is for mesenchymal stem cells paracrine function and cell to cell contact.
I don’t know who to reply too….but //I am 56 and broke my back at 13-gymnast) L3 L4 L5… continued training and got through the pain and kinda went away…. then at 54 woke up. in so much pain and could not walk at all…OMG …I have trained all my life..running..lifting ..biking …pilaties..yoga..every day …. then I found stem cell which I study for many years..and found a doctor. had it done for the 2nd time from 2 years ago and I am bionic!! NO PAIN!! now doing my knee as well. I work as a medical aesthetician and we use stem cell from South Korea and needle it in the skin …strech marks ..GONE !!! wrinkles gone tight young skin .. I will forever use stem cell and was wondering about repair for organs….just was talking about it with my regenerative doctor …. no more surgery for repair or recover it can be done with stem cell, I have umbilicord in my back… and its like I am 30 THANK SCIENCE AND THE REGENERATIVE DOCS!!!
Elizabeth, I’d love to talk to you to hear more about your experience if you don’t mind. I know you wrote this a few years ago but I am now researching & looking for the best stem cell treatment for a degenerative back.